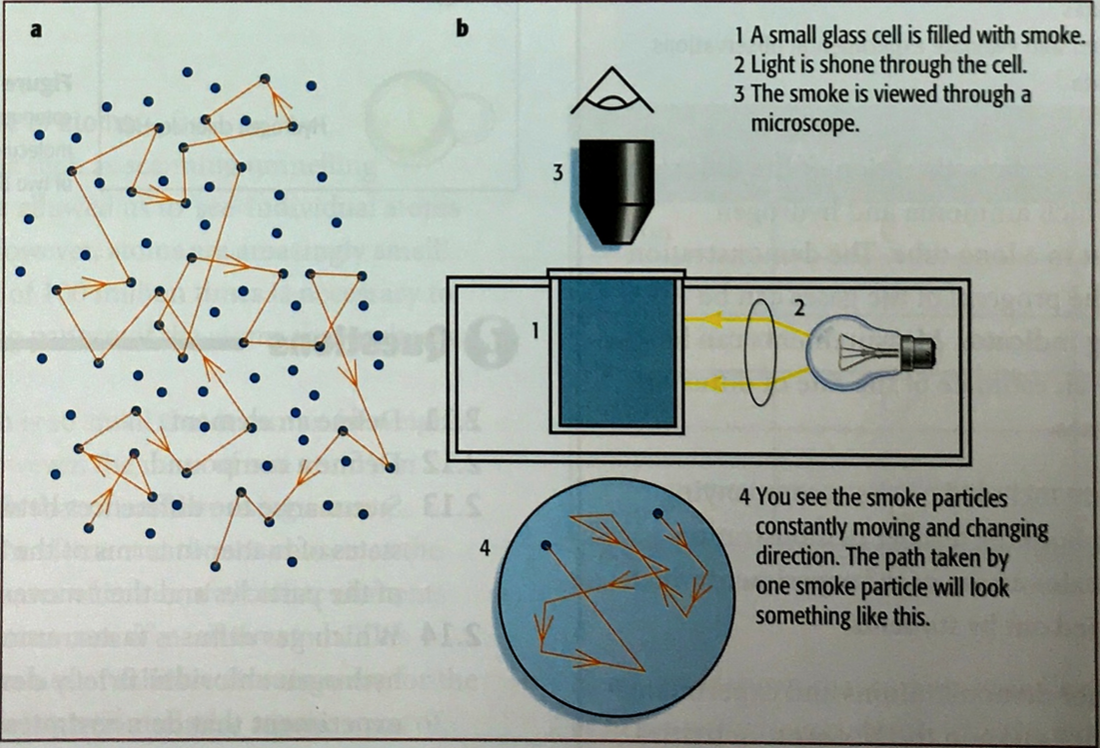
Show an understanding of the random motion of particles in a suspension (sometimes known as Brownian motion) as evidence for the kinetic particle (atoms, molecules or ions) model of matter
Particles in a fluid (liquid or gas) will move around in random directions and collide with each other and their surroundings. When they collide they change direction. this random movement will cause them to spread out or diffuse through a liquid, this can be observed through a colour change or by a reaction occurring where two substances meet.
Describe and explain diffusion
The idea that fluids (liquids and gases) are made of moving particles helps us to explain what is happening in diffusion. There a few examples of diffusion that you should be familiar with;
Describe and explain Brownian motion in terms of random molecular bombardment
State evidence for Brownian motion
|
There are several examples of experiments where you can directly observe the effects of brownian motion. Einstein was the first to notice the effect of random bombardment of water molecules against the much larger pollen grains causing them to move around randomly.
You can see the same effect when you observe smoke particles under a microscope. They will move around randomly caused by their collisions with smaller unobservable particles. |