Define oxidation and reduction in terms of oxygen loss/gain.
|
Oxidation
In terms of oxygen; oxidation is when an atom or ion gains oxygen. Reduction In terms of oxygen; reduction is the when an atom or ion (a species) loses oxygen. Combustion Combustion is the reaction of a substance with oxygen causing the release of energy. The reaction is exothermic and often involves a flame.
|
|
Rancidity
Oxidation can also have a damaging affect on food. When fats and oils in butter and margarine are oxidised they become rancid. Their taste and smell change and become unpleasant. This is why manufacturers may add chemicals 'antioxidants' to prevent oxidation. Keeping food at low temperatures (in a fridge) will also slow down oxidation, or reducing the amount of oxidation by storing food in an air tight container. Some foods such as crisps are even stored in other gases such as pure nitrogen to prevent oxidation. |
|
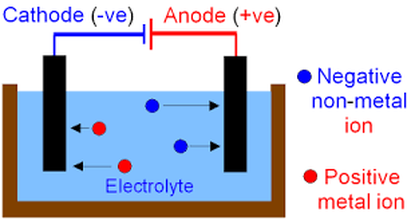
Electrolysis
This is a process where an electric current is used to drive a reaction. This is also a redox reaction. At the positive electrode(called the anode) negative ions lose their electrons which means they are oxidised whilst at the negative electrode (cathode) positive ions gain electrons which means they are reduced. |
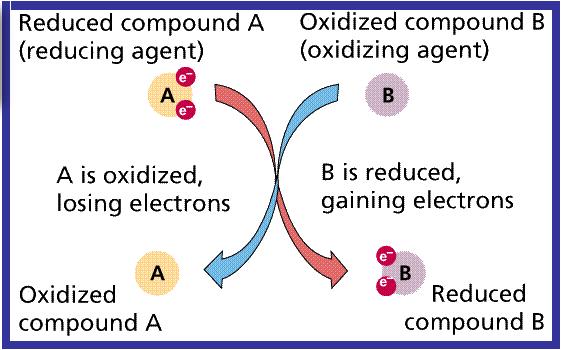
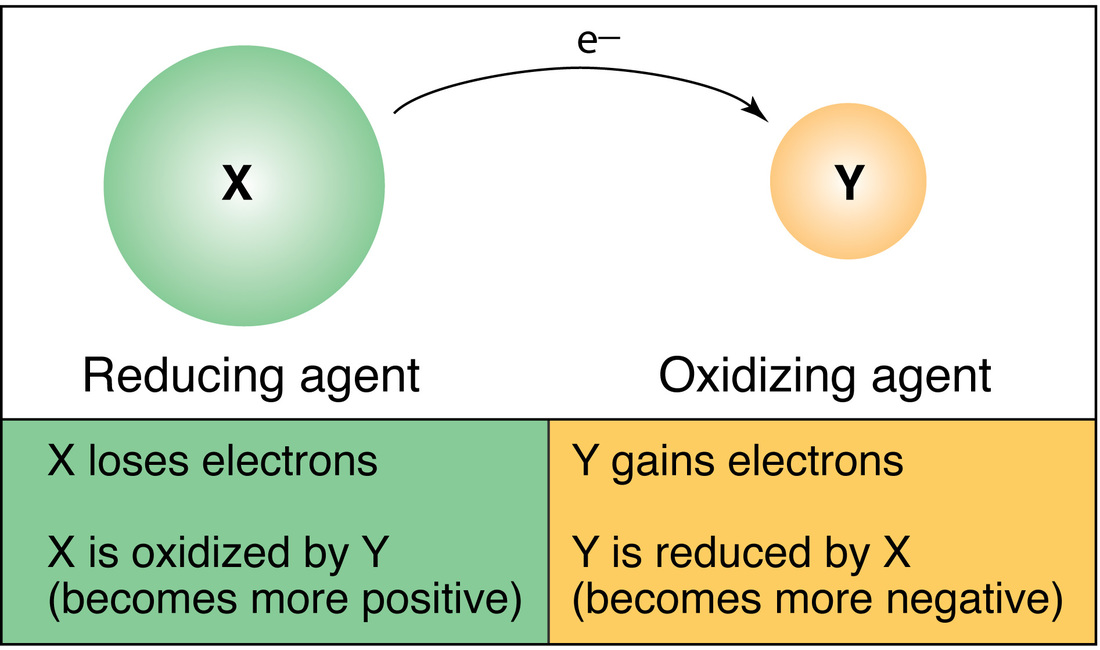
Define redox in terms of electron transfer
Reduction
Reduction is the when an atom or ion (a species) GAINs electrons. Oxidation Oxidation is when an atom or ion LOSES electrons. A Redox reaction Involves a transfer of electrons from one species to another. The way to remember which is which is: OIL RIG Oxidation is loss (of electrons) Reduction is gain (of electrons |
|
Copper (II) sulfate
Copper (I) oxide
|
|
|
Using potassium iodide to test for an oxidising agent
Potassium Iodide (KI) can be very useful because when it is oxidised a colour change is produced. The Iodide ion (I-) is oxidised to iodine(I2). The colour of the solution will change from colourless to yellow-brown. If you then add starch indicator it will turn blue-black. |
|
potassium dichromate(VI)
carbon
|
Define oxidising agent as a substance which oxidises another substance during a redox reaction. Define reducing agent as a substance which reduces another substance during a redox reaction.Oxidising Agent
A substance that will add oxygen to another substance. The commonest oxidising agents are oxygen (or air), hydrogen peroxide, potassium manganate(VII) and potassium dichromate(VI). Reducing Agent An element or compound that will remove oxygen from other substances. The most common reducing agents are hydrogen, carbon and carbon monoxide. Remember that, in the process of acting as a reducing agent, that substance will itself be oxidised. The reducing agent will gain the oxygen it is removing from the other compound. The reverse is also true for an oxidising agent. |
Identify oxidising agents and reducing agents from simple equations
Consider the reaction:
2Cu + O2 --> 2CuO
It is clear that copper has been oxidised, but what has been reduced? The copper atoms have lost electrons to become Cu2+ ions. The oxygen molecules in the gas have split and become O2- ions in the copper oxide. The oxygen atoms have gained electrons in order to become negative (remember atoms in an element are always neutrally charged). And so we can say the oxygen atoms were reduced. The copper must be a reducing agent.
Consider the displacement reaction:
Zn + CuSO4 --> ZnSO4 +Cu
Now it is not so clear, the solid Zinc metal has turned into Zn2+ ions meaning that it has lost electrons to the copper, loss of electrons is oxidation so the copper must be the Oxidising agent. The Copper ions have gained electrons to change in to Cu atoms from Cu2+ ions, this means that the Zinc must be the Reducing agent.
Finally consider the displacement of Iodide ions from potassium iodide solution by chlorine gas.
Cl2(g) + 2I-(aq) --> 2Cl-(aq) + I2(aq)
The oxidation state of the chlorine atoms is 0 because they are an element and have not yet lost or gained electrons, whereas the oxidation state of the chloride ions at the end are -1 because they have gained one electron per atom, this decrease in oxidation state is called reduction meaning that the chlorine must be the oxidising agent. Using the reverse logic we know the Iodide ions are the reducing agent for the chlorine.
2Cu + O2 --> 2CuO
It is clear that copper has been oxidised, but what has been reduced? The copper atoms have lost electrons to become Cu2+ ions. The oxygen molecules in the gas have split and become O2- ions in the copper oxide. The oxygen atoms have gained electrons in order to become negative (remember atoms in an element are always neutrally charged). And so we can say the oxygen atoms were reduced. The copper must be a reducing agent.
Consider the displacement reaction:
Zn + CuSO4 --> ZnSO4 +Cu
Now it is not so clear, the solid Zinc metal has turned into Zn2+ ions meaning that it has lost electrons to the copper, loss of electrons is oxidation so the copper must be the Oxidising agent. The Copper ions have gained electrons to change in to Cu atoms from Cu2+ ions, this means that the Zinc must be the Reducing agent.
Finally consider the displacement of Iodide ions from potassium iodide solution by chlorine gas.
Cl2(g) + 2I-(aq) --> 2Cl-(aq) + I2(aq)
The oxidation state of the chlorine atoms is 0 because they are an element and have not yet lost or gained electrons, whereas the oxidation state of the chloride ions at the end are -1 because they have gained one electron per atom, this decrease in oxidation state is called reduction meaning that the chlorine must be the oxidising agent. Using the reverse logic we know the Iodide ions are the reducing agent for the chlorine.
Home Study Tasks
|
| ||||