Describe and explain the effect of concentration, particle size, catalysts (including enzymes) and temperature on the rate of reactions
Describe the application of the above factors to the danger of explosive combustion with fine powders (e.g. flour mills) and gases (e.g. methane in mines)
Demonstrate knowledge and understanding of a practical method for investigating the rate of a reaction involving gas evolution
Devise and evaluate a suitable method for investigating the effect of a given variable on the rate of a reaction
Interpret data obtained from experiments concerned with rate of reaction
Describe and explain the effects of temperature and concentration in terms of collisions between reacting particles (An increase in temperature causes an increase in collision rate and more of the colliding molecules have sufficient energy (activation energy) to react whereas an increase in concentration only causes an increase in collision rate.)
Describe and explain the role of light in photochemical reactions and the effect of light on the rate of these reactions (This should be linked to section 14.4.)
Heat is not the only form of energy that can break bonds and start chemical reactions. Some chemical reactions are affected by light energy, these are called photochemical reactions. Like heat light will speed up some chemical reactions and provide the activation energy needed for them to begin.
Describe the use of silver salts in photography as a process of reduction of silver ions to silver;
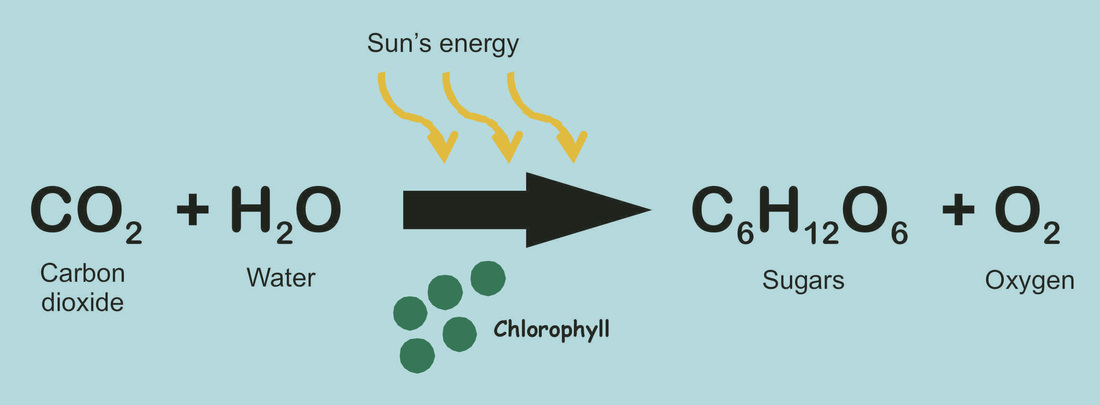
and photosynthesis as the reaction between carbon dioxide and water in the presence of chlorophyll and sunlight (energy) to produce glucose and oxygen
|
Photosynthesis
Photosynthesis traps energy when sunlight falls on leaves containing the green pigment chlorophyll, the reaction converts water and carbon dioxide into glucose and oxygen. the plant will then use the glucose to make starch and other carbohydrates. Photosynthesis is the reverse reaction of that which occurs in all living things; respiration. The energy that is stored by plants in this process is called biomass energy. |
|
Photography
Silver salts also have a photochemical reaction, when exposed to light they get darker. The darkening is caused by the salts decomposing into their constituent elements. For example:
2AgBr --> 2Ag +Br2 This is also a redox reaction because the bromide ions lose electrons and the silver ions gain electrons. This photochemical reaction was the basis of all black and white and colour photography. Photographic film was made from a flexible plastic coated in a light sensitive emulsion of silver salts. Where the film is exposed to the most light the silver solid is precipitated out and so darkens the film. This was called the negative image and they would need to be used to create the final image as they would be a reversal of the true image.
|
Substitution reaction of Alkanes with Chlorine
|
This links into what you will learn later with organic chemistry. Simply put when exposed to the ultraviolet light in sunlight chlorine molecules will split into separate energised atoms.
These atoms will then readily react with methane (CH4so the overall reaction is that the chlorine atom replaces (substitutes) one of the hydrogens on the methane molecule to gove chloromethane (CH3Cl). With lots of chlorine this reaction can repeat until all of the hydrogens have been substituted giving the product of tetrachloromethane (CCl4). You may have heard of some of the chemicals formed in this way; trichloromethane is commonly known as chloroform which was an early anaesthetic, another chemical made in the same way, 1,1,1-trichloroethane, is the solvent used in dry-cleaning. |
Check Understanding
Home Study Tasks
|
|
|
|
| ||||||||||